 
During this experiment, electrons are accelerated through a low-pressure gas, and the collisions between the electrons and gas atoms provide enough energy for the electrons in the gas atoms to change from one state to another. The Bohr model of the atom has been supported by a number of experiments since 1914, including the Franck-Hertz experiment. Planck’s formula correctly describes how radiation from heated bodies is determined. 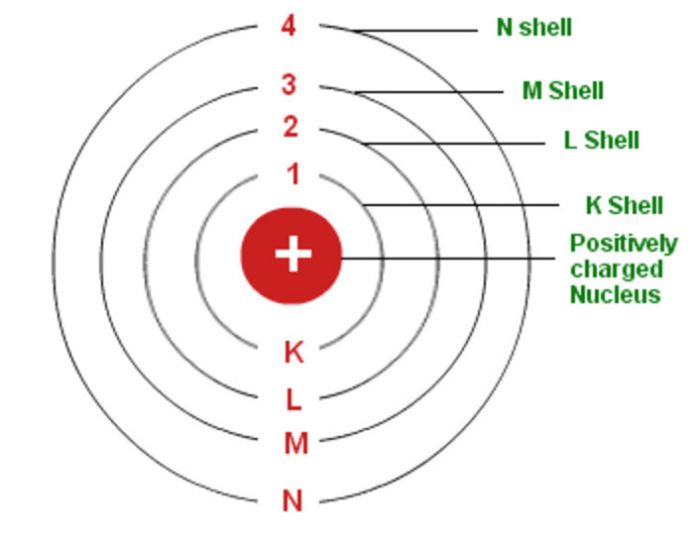
The angular momentum of an electron, according to Bohr, is quantized and can only be measured in discrete values. Planck’s constant is an action principle with dimensions of energy, time, momentum, length, or angular momentum. The Rutherford model‘s design was modified by Bohr by requiring that electrons orbit fixed orbits of fixed sizes and energy. Charged particles moving on curved surfaces emit electromagnetic radiation, according to classical mechanics. In 1913, Bohr proposed a quantized shell model of an atom to explain how electrons can have stable orbits around the nucleus. This model revolutionised the way we think about atoms and laid the foundation for the development of quantum mechanics. He also proposed that the electrons could only occupy certain specific orbits, and that when electrons jumped from one orbit to another, they emitted or absorbed electromagnetic radiation. Bohr’s model proposed that atoms are composed of a nucleus surrounded by electrons that orbit the nucleus in circular paths. This model was the first to accurately explain the structure of atoms, which had been a great scientific mystery up until then. Niels Bohr was a Danish physicist who developed a revolutionary model of the atom in 1913. This model is used by physicists to explain a wide range of nuclear phenomena, allowing us to rethink our understanding of atomic structure. Electrons move through circles in circles at various speeds depending on the state of the element, which has its own set of shells. These diagrams, which show electron images orbiting the nucleus like planets, provide a visual representation of this model. This model describes electrons traveling through distinct circular orbits, or shells, around the nucleus of atoms as a result of electron traveling through distinct circular orbits. The Bohr Model, created by physicist Niels Bohr in 1913, is widely regarded as the foundation of modern physics. Unlocking The Secrets Of Atomic Structure: The Bohr Model The electron could enter three-dimensional space in Schr inger’s model. The size of the orbit, which was described by the n quantum number, was the most important information. The Bohr model defined the electron distribution in one way: it used one quantum number to describe its movement. Despite the incorrect prediction of the Bohr’s model for the hydrogen atom, its ground state has no zero orbital angular momentum. This is why Bohr’s theory wasn’t as accurate as it should have been in 1913. It also failed to explain the Stark effect and Heisenberg Uncertainty Principle, despite having a clear understanding of them. When an electron enters one of these orbits, it has its energy fixed.īohr’s Atomic Model Theory does not explain why magnetic fields have an effect on the wavelengths of atoms. The electron encircles the nucleus of the atom in orbital paths specified in the Bohr model, also known as a planetary model. In this article, we’ll explore how Bohr’s 3D model of the atom worked and how it was used to further our understanding of atomic structure. This 3D model was based on the same principles as the 2D version but was more visually appealing and allowed for a more comprehensive analysis of the atom’s structure. While the Bohr model was initially a 2D model, Bohr later went on to create a three-dimensional version of the atom. But did Bohr ever make a 3D model of the atom? The answer is yes. This model had a profound impact on scientific understanding of the atomic structure. He is best known for proposing the Bohr model, which is a model of the atom. Niels Bohr was a Danish physicist who made significant contributions to the field of atomic theory and quantum mechanics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
